Portfolio
We at BiOZEEN practically touch upon every unit operation of the Biopharmaceutical manufacturing value chain with:
- Design & Build of customized Fermentors, Bioreactors and other Bioprocess equipment
- Value Added Integration Services which integrates process, engineering, automation and facility design, with regulatory expertise to support each phase of the project life cycle
- Training to enable industry professionals reduce rejects and increase yield, and to develop industry ready academia
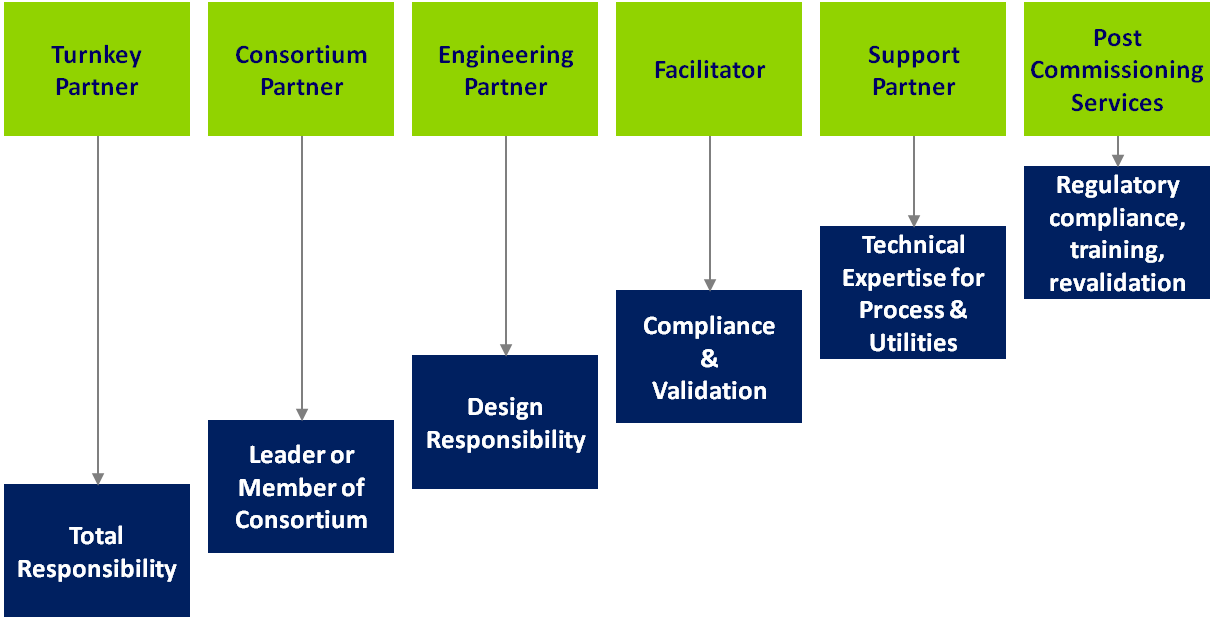
BiOZEEN project model is tailored to meet industry needs at every stage of the project life cycle including requirement of:
- Product Support with filter validation & method development services , in compliance with the guidelines of international bodies such as US FDA, EU GMP, EMEA, to name a few, also encompassing requirements listed in PDA TR # 26 (rev. Sep 2008), enabling you release the molecule fast & first to the market
- Process Support provided by bioprocess solutions, with core capability in biologics/ biopharmaceutical processing, to ensure operational efficiency, process integrity and optimized throughput – all in compliance with the regulatory guidelines
- Production Facility Support
- cGMP Facility Design Solutions
- Design & Build of customized Fermentors, Bioreactors and allied upstream equipment to suit the process, facility and regulatory guidelines requirements. At BiOZEEN, we can also support your equipment rehabilitation, retrofit and new customized equipment requirements. Our Automation Team at BiOZEEN ensures user friendly operations which are 21 CFR Part 11 ready, in the highly challenging Pharma & Biopharma production space. Hence enabling you handle solutions & processes with a touch of simplicity.
- People Support with Training that practically educates professionals enabling them reduce rejects and increase yield.
This scope of services makes us a single source solution provider to execute product, process and production facility engineering for your projects.
To obtain more information, you could connect with us by way of our Contact Details or fill in our Enquiry Form and we will touch base with you shortly.